|
| |
Using the QuickScreen™ Pro Drug Scan Test
TIMING
DEVICE
|

|
The QuickScreen Pro Multi-Drug Test Card now incorporates an automatic timing device. The timer has two indicator "lights" that will turn a red color tone as critical periods are reached during the development of test panel results. A "result ready" indicator will show when test results are completely developed and ready to be interpreted, while a "result expired" indicator will show when test development time is beyond that specified for a correct interpretation of test results. This new timing device eliminates the need for a separate timer during testing and potential interpretation errors due to improper waiting periods.
|
SPECIMEN
COLLECTION
The QuickScreen Pro Multi-Drug Test Card is formulated for use with human urine specimens only. Fresh urine does not require any special handling or pretreatment. Urine samples should be collected such that testing may be performed as soon as possible after the specimen collection, preferably during the same day. The specimen may be refrigerated at 2-8° C for 2 days or frozen at -20° C for a longer period of time. Specimens that have been refrigerated must be equilibrated to room temperature prior to testing. Specimens previously frozen must be thawed, equilibrated to room temperature, and mixed thoroughly prior to testing.
PRECAUTIONS
Urine
specimens may be potentially infectious. Proper handling and disposal methods should be
established. Avoid cross-contamination of urine samples by using a new specimen collection
container for each urine sample.
Note: Urine
specimens, and all materials coming in contact with
them, should be handled and disposed as if capable of transmitting infection. Avoid
contact with skin by wearing gloves and proper attire.
TEST PROCEDURE
INTERPRETATION
OF RESULTS
|
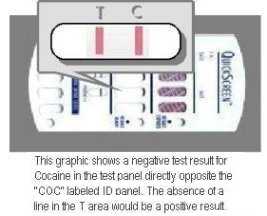
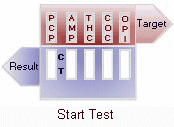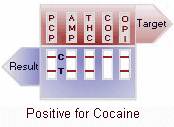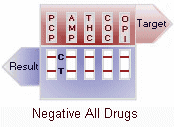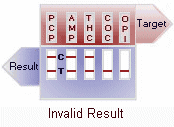
(-) Negative: Two colored lines adjacent to each drug name should be observed in the viewing window. The line in the test region (T) is the drug probe line; the line in the control region (C) is the control line, which is used to indicate proper performance of the device. The test line intensity may be weaker or stronger than that of the control line.
(+) Positive: Only one colored line appears in the control region (C). The absence of a test line indicates a positive result for that drug.
(-/+) Inconclusive: A faint*, indistinguishable line appears in the test region (T) . While probably suggesting a negative result, the test is inconclusive and suggests that the drug in the panel could be near the cut-off concentration level for detection. Perform a second test or take the sample for more extensive laboratory testing. Because of the quality controls built into the test device, an inconclusive result is highly unlikely.
Invalid: No line appears in the control region. Under no circumstances should a positive sample be identified until the control (C) line forms in the viewing area. If the control line (C) does not form, the test result is invalid and the assay should be repeated.
View
Result Graphic |
|
*Note: An
extremely faint line (barely visible) in the test region indicates that a drug
metabolite in the sample may near the cut-off concentration level for the test. Normally,
any line in the test area should be interpreted as a negative test. If suspicion
or uncertainty remains regarding the test results, there are several options
available: The donor can be retested at another time with a new test card and
sample, the same sample can be retested using a new test card, or the sample can
be evaluated by a testing laboratory. If laboratory confirmation is desired or
indicated, please see the QuickScreen Pro Drug Test
Kit
LIMITATIONS
OF TEST PROCEDURE
-
This
assay is designed for use with human urine only.
-
A positive result with any of the tests indicates only the presence of a
drug/metabolite and does not indicate or measure the level of intoxication.
-
There is a possibility that technical or procedural errors as well other
substances as factors not listed may interfere with the test and cause false results.
Included with each card are lists of substances that could produce positive results, or
that are known not to interfere with test performance.
-
If
it is suspected that the samples have been mislabeled or tampered with, a new specimen
should be collected and the test repeated or an
Adulteration ID Test should be obtained.
For details on the chemistry technology of the test please refer to
test technology
or the Drug FAQ page.
|